Need to Launch a Companion Diagnostic (CDx) in China?
Speed up your time-to-market by partnering with Novodiax

China presents a significant pharmaceutical market opportunity, with 4.2 million new cases of cancer and 2.8 million resulting deaths in 2018 alone. Despite the strong need for advanced antibody-based cancer therapies and companion diagnostics (CDx), many companies must delay drug introduction whilst country of origin approval is obtained for the CDx.
Novodiax is a proven in vitro diagnostics (IVD) and immunohistochemistry (IHC) leader with in-country partners and a track record of success in China, and we’d like to collaborate with you to bring your innovations to market more rapidly.
Novodiax can help you get to market 6-12 months faster
Manufacturing
Don’t waste valuable time and money waiting for country of origin approvals! Get to market with your CDx sooner by manufacturing in-country at our state-approved GMP facility in Southwestern China.
Commercialization
Our commercial and administrative teams, based in the business hub of Shanghai, have a successful track record of bringing in vitro diagnostic (IVD) IHC products to market and selling to the largest hospitals in China.
Leverage our revolutionary ihcDirect® rapid IHC technology
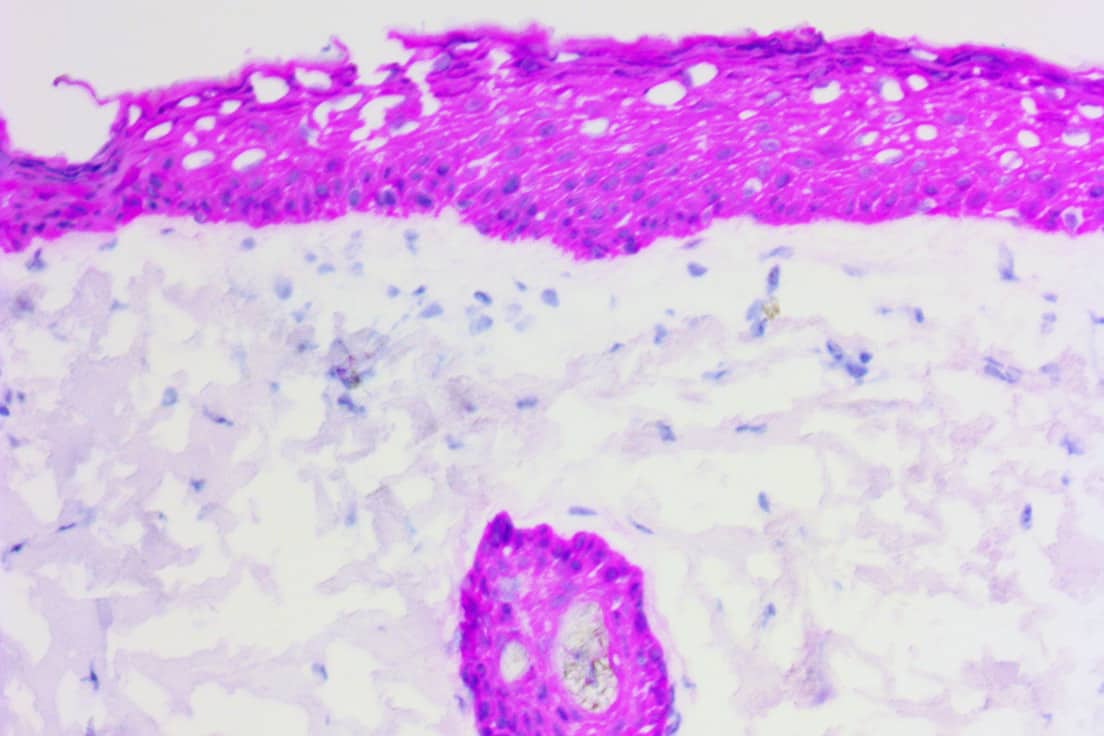
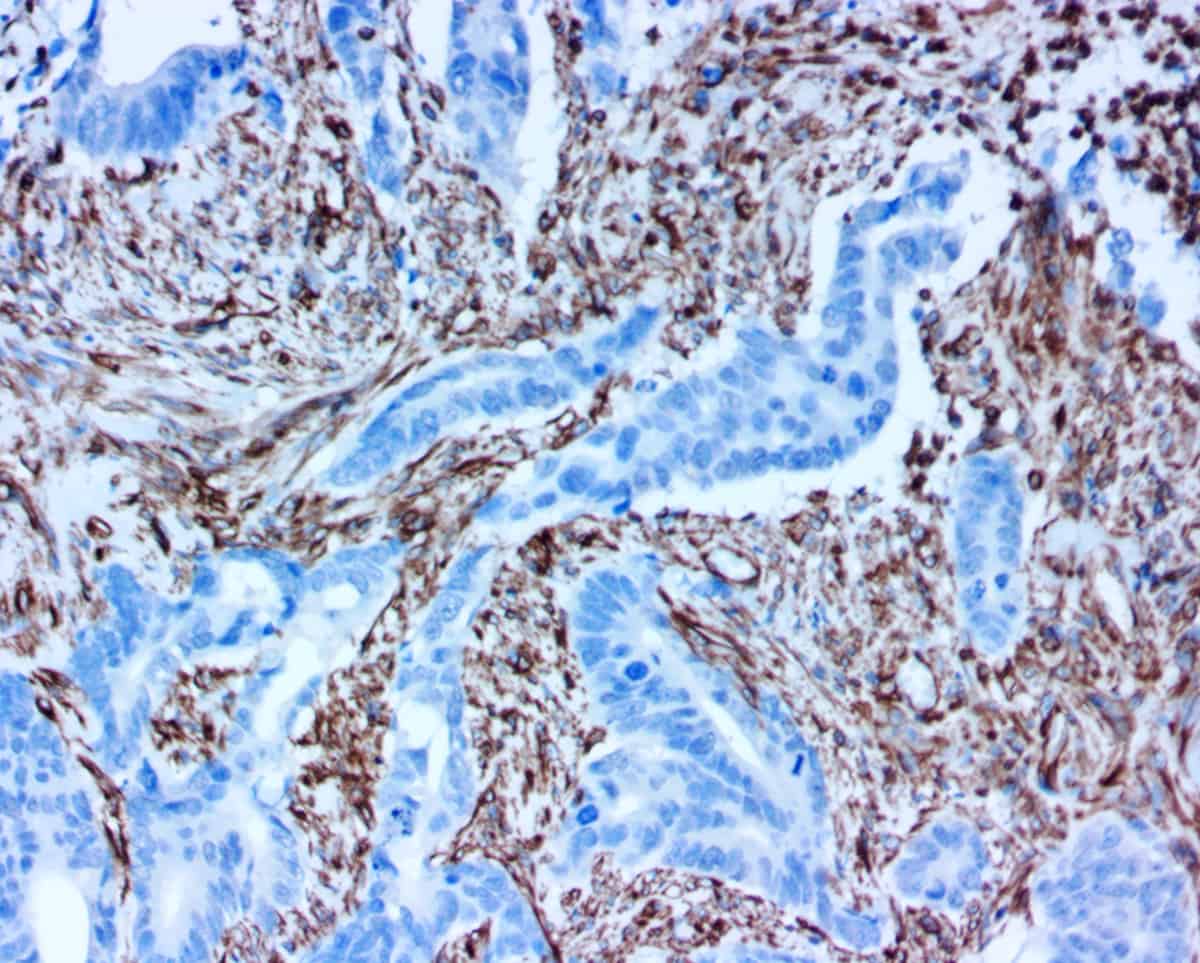
Using the ihcDirect rapid IHC technology, Novodiax can conjugate your antibody-of-choice to our proprietary polyHRP (polymerized horseradish peroxidase) signal amplification technology, producing a sensitive assay that is perfect for rapid, high-precision IHC testing on frozen tissue. [Click on the adjacent poster to read about the potential use of ihcDirect assays in a companion diagnostic for Herceptin-based treatment of metastatic breast cancer]. ihcDirect assays can be run on a variety of open automated platforms (e.g. Leica, Thermo, etc.)
Even if you’re at an earlier stage with your CDx, Novodiax can help accelerate CDx development
Leverage our revolutionary ihcDirect® rapid IHC technology to accelerate development and testing of your CDx. Our technology can be used to speed up biomarker identification, clone selection, validation and profiling, tissue cross reactivity and toxicity testing, etc. We can provide custom support for your specific needs, or even partner with you throughout the entire development process.
LET’S DISCUSS PARTNERSHIP OPPORTUNITIES
If you are interested in collaborating with an experienced IVD IHC technology leader who can help you get to market in China 6-12 months faster, we would love to discuss partnership opportunities with you.