The following article discusses the results of a clinical study that was conducted by Mei Liu et al. at the PLA General Hospital and published in Discovery Medicine (July 2019). Read the original publication here: http://www.discoverymedicine.com/Mei-Liu/2019/07/direct-immunohistochemistry-ihc-improves-intraoperative-diagnosis-of-breast-cancer/
Frozen sections have been used for intra-operative assessment of breast lesions for more than a century. These rapid, easy-to-perform tests can help physicians:
- Confirm a pathological diagnosis (when pre-operative biopsies are inconclusive or not feasible),
- Assess resection margins (e.g. during a lumpectomy procedure), and
- Detect occult metastases (by way of sentinel lymph node analysis).
… all while the patient is still in the room.
In turn that can mean fewer surgeries, lower recurrence rates, reduced patient anxiety, better use of physicians’ time, and a host of other benefits for patients, providers, and health care institutions.
At times, frozen section hematoxylin and eosin (H&E) stains can be unclear. Here are three scenarios where morphology alone is insufficient for clinical decision making:
- Assessing cell populations that look similar under a microscope,
- Differentiating tumors from non-cancerous inflammation (e.g. reactive mesothelial cells), and
- When histological morphology has become obscured by freeze artifacts or mechanical/ thermal damage.
In such cases, rapid immunoassays on frozen sections can provide a valuable second opinion to immediately aid physicians in their decision making.
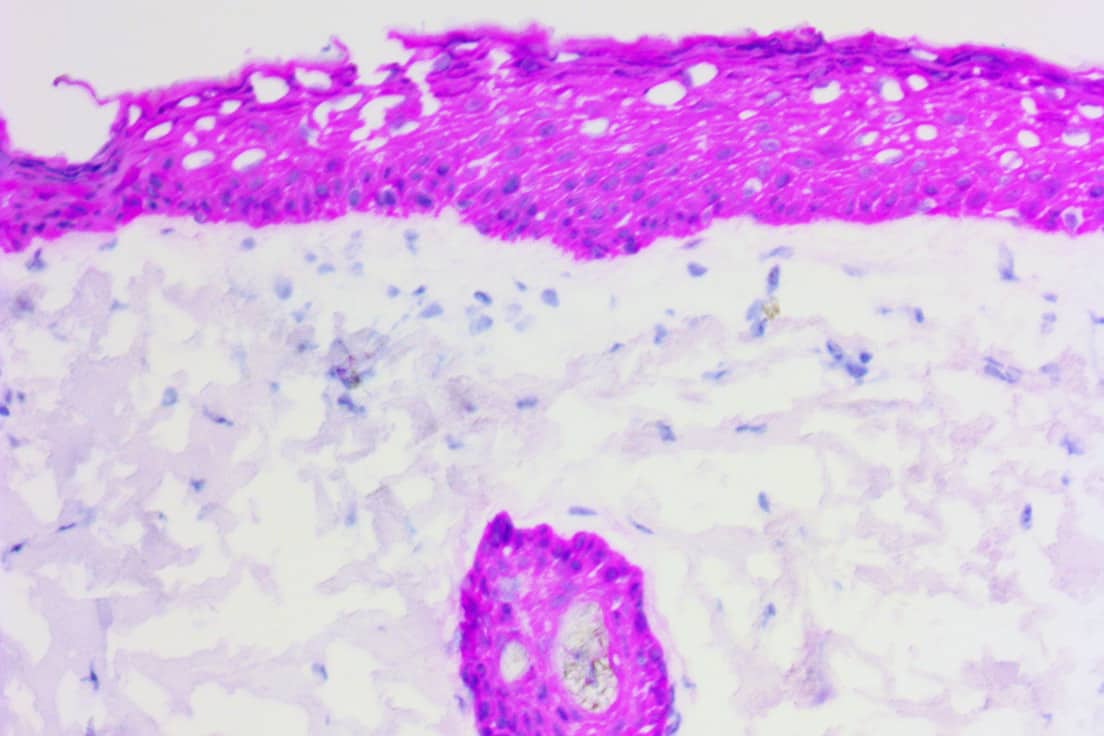
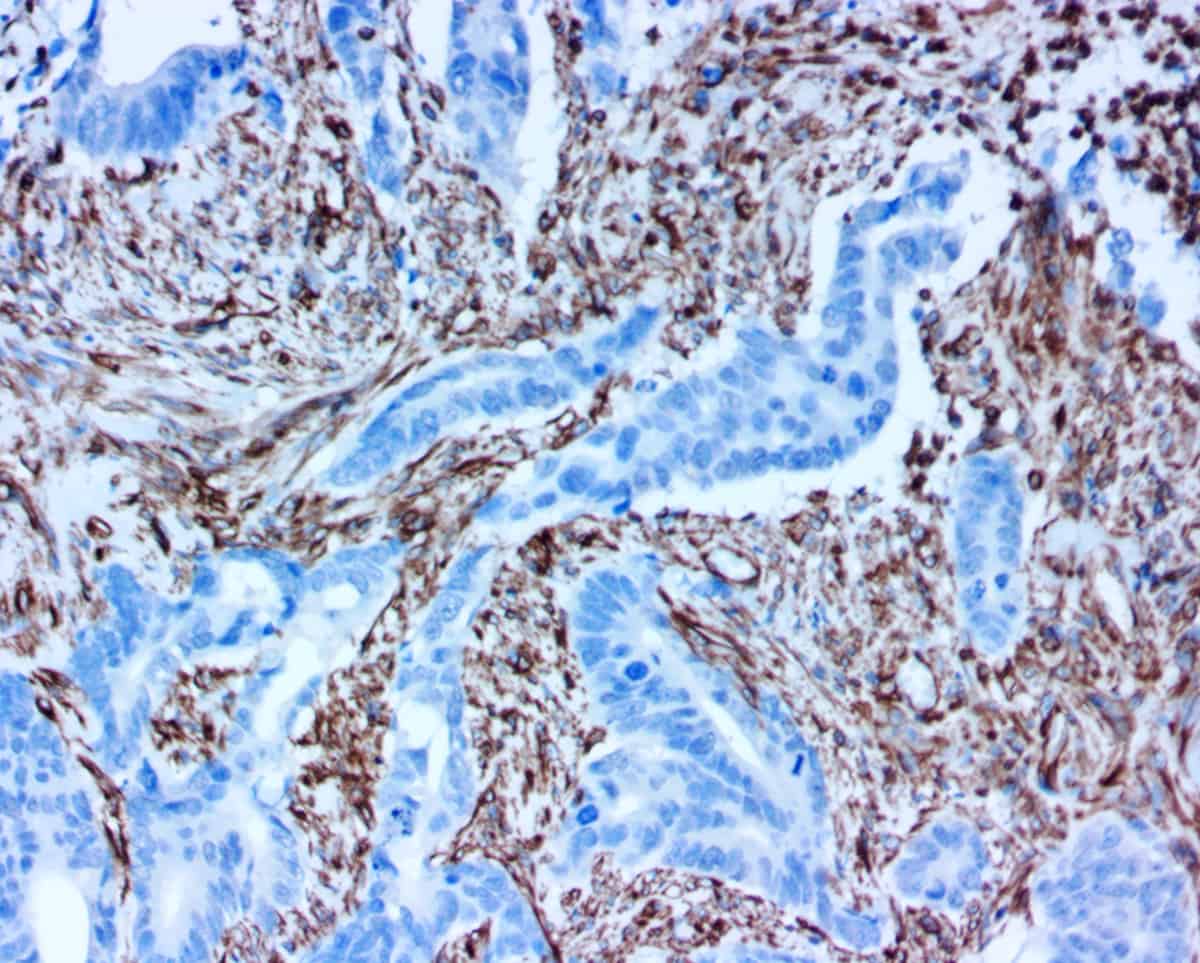
For instance, smooth muscle myosin heavy chain (SMMHC) is a myoepithelium-associated protein that has been shown to help with distinguishing carcinoma in situ from invasive breast cancer1. Negative staining with the SMMHC antibody indicates a lack of myoepithelial cells (which are thought to act as tumor suppressors), and therefore potential infiltration of tumors into adjacent spaces. Cytokeratins have been shown to be helpful in distinguishing benign usual ductal hyperplasia (UDH) – characterized by high expression of high molecular weight cytokeratins (HMWCK) such as CK5 – from atypical ductal hyperplasia (ADH) and ductal carcinoma in situ (DCIS) – characterized by low or weak expression of HMWCK1.
Unfortunately, however, many immunohistochemistry (IHC) tests on the market today are impractically slow for intra-operative frozen section IHC analysis. Some IHC products require lengthy antigen retrieval steps, incubations of one or more antibodies and detection polymers, and (in the case of formalin-fixed, paraffin-embedded or FFPE sections) hours-or-days-long perfusion of tissues with formalin. Also, many IHC products fail to perform well on frozen sections, as the vast majority of IHC companies focus on optimizing their products for FFPE sections only. Thus, IHC is often ruled out as an impractical choice for intra-operative testing. That is, until now.
Researchers at the PLA General Hospital (Beijing, China) conducted a study to determine whether IHC staining could be performed fast enough to be useful in an intra-operative setting. To accomplish this, they utilized Novodiax ihcDirect® assays – specifically the Cytokeratin 5 (CK5) and SMMS-1 (SMMHC) antibodies – which are among the fastest direct IHC products available for in vitro diagnostic (IVD) use. They also looked at whether intra-operative IHC truly provides enhanced value when used in combination with H&E (compared to H&E alone).
Details of the Study2
Location: PLA General Hospital (Beijing, China)
Dates: August 2015 to May 2017
Study Population: 97 patients, ages 22 to 80 (median 46), who underwent either breast mass resection, segmental mastectomy, or simple mastectomy
Specimens: 102 tissue samples (5 of the patients had bilateral lesions, producing two specimens each instead of one)
Procedure: Four frozen section slides were produced from each lesion. Two of the slides were routinely stained with H&E and analyzed by a pathologist. The other two slides were stained with Cytokeratin 5 (CK5) and smooth muscle myosin heavy chain (SMMHC) antibodies, and they were analyzed by a different pathologist who read them in combination with the H&E slides. All four frozen sections were compared to IHC stained FFPE slides, which served as the gold standard for final diagnosis.
The researchers found that “the entire procedure of direct IHC can be finished within 10 minutes […] [making it] feasible for the use of intra-operative frozen section diagnosis”. Further, the IHC frozen sections produced “little or no background stains that are comparable to the conventional IHC stains on FFPE tissues”.
This combination of speed, sensitivity, and specificity is made possible by the proprietary ihcDirect polymerized horseradish peroxidase (polyHRP) technology utilized in Novodiax products.
Not only that, the rapid IHC tests were also shown to help significantly improve the accuracy of intra-operative assessment of breast lesions.
For instance, when read alone without IHC, the H&E frozen sections resulted in 27 lesions being categorized as papilloma with UDH. However, according to the gold-standard IHC on FFPE tissues, only 14 of those diagnoses were correct (51.9%). In fact, the FFPE sections produced 31 total diagnoses of papilloma with UDH. Six of the misdiagnosed lesions were actually papilloma with no hyperplasia, three were papilloma with ductal carcinoma in situ (DCIS), two were papilloma with atypical ductal hyperplasia (ADH), and one was encapsulated papillary carcinoma. On the other hand, when reading frozen section IHC in combination with H&E, the correct diagnoses was made in all of the papilloma with UDH cases.
Overall, compared to H&E alone, frozen section IHC + H&E resulted in an:
- Increase in diagnostic accuracy, from 70% to 97%
- Decrease in false-negative diagnoses, from 53% to 0%
- Decrease in deferred diagnosis rate, from 21% to 3%
Breast cancer is the most common cancer in women worldwide, accounting for 25.4% of all new cases diagnosed in 2018, and there is a serious need for innovative approaches to diagnosis and treatment. If you would like to learn more about Novodiax ihcDirect products and incorporating rapid intra-operative IHC testing into your practice, please contact us at Novodiax today.
To read the original paper published in Discovery Medicine, please visit: http://www.discoverymedicine.com/Mei-Liu/2019/07/direct-immunohistochemistry-ihc-improves-intraoperative-diagnosis-of-breast-cancer/
[1] Liu, Haiyan. Application of immunohistochemistry in breast pathology a review and update. Arch Pathol Lab Med. Vol 138:1629-1642. Dec 2014.
[2] Liu, Mei et al. A Direct Immunohistochemistry (IHC) Method Improves the Intra-operative Diagnosis of Breast Papillary Lesions Including Breast Cancer. Discov Med 28(151):29-37, July 2019.